
The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).Ītomic Structure / isotopes atoms of some elements have two or more different atomic masses,called isotopes.Ītomic Structure Atomic weight: Weighted average of the atomic masses of the atom’s naturally occurring isotopes. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of electrons in a neutral atom is equal to the number of protons. massnumber:Ě = Z + N number of protonsis the same for all atoms of a given element, number of neutrons (N) maybe variable.Ītomic Structure The number of protons in the nucleus of the atom is equal to the atomic number (Z). Zranges from 1 for hydrogen to 92 for uranium (thehighestforthenaturallyoccuringelements).Ītomic Structure The atomic mass (A) of a specific atom: the sum of thenumber of protons and neutrons within the nucleus.Atoms with the same atomic number have the samechemical properties and belong to the sameelement.For an electrically neutral atom, atomic number = number of electrons.Every different atom has a characteristic number ofprotons in the nucleus.Toexplainthelinespectra, Bohrproposedthatelectrons of specificenergymoved in circularorbitsaroundthenucleusandcould not existbetweentheseorbits.Ītomic Structure Atomsare composed of protons– positively charged particles neutrons– neutral particles electrons– negatively charged particles in orbitals surrounding the nucleus. 1922 Nobel prize in physicsīohr’satom Rutherford’s model predicted a rainbow of colorsratherthandiscretelinesobtainedfrom an atomiclinespectra. With each orbit only able to contain a set number of electrons. 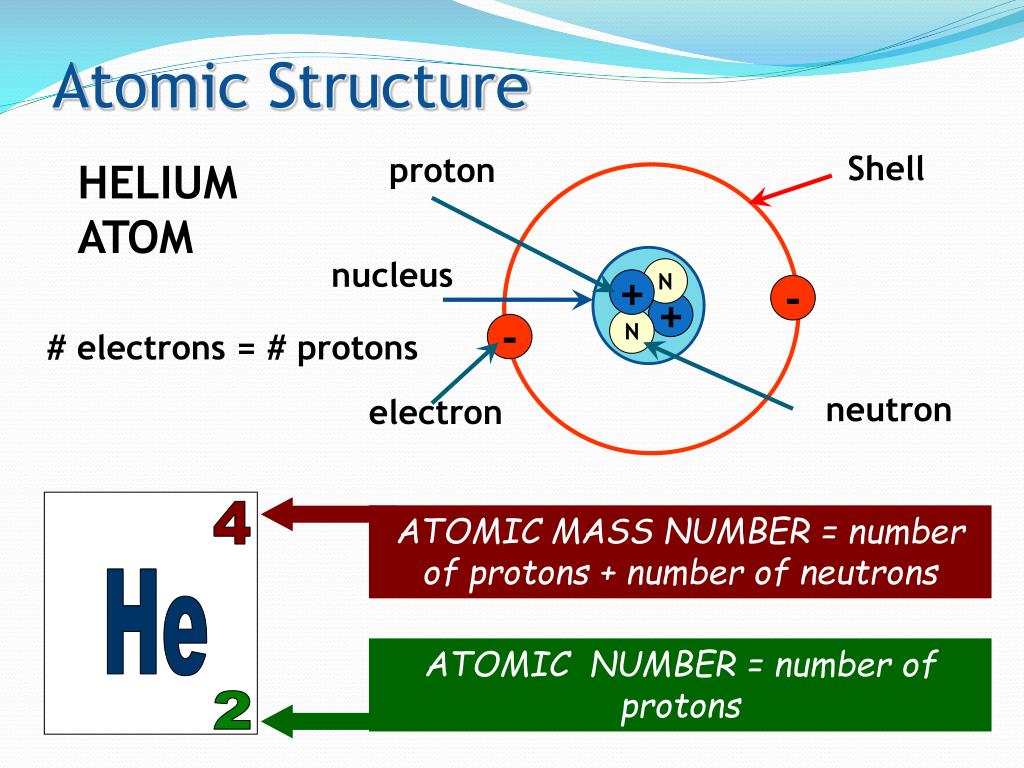
Bohr refined Rutherford's idea by adding that the electrons were in orbits. History of the atom NielsBohr / Danish / a footballfanatic 1913 studied under Rutherford at the Victoria University in Manchester. With this holding the electrons in place by electrical attraction 1908 Nobel prize in Chemistry History of the atom 1910 Ernest Rutherford / Cambridge student of Thompson proposeda more detailed model with a central nucleus: positive charge was all in a central nucleus. History of the atom Joseph John Thompson / Cambridge 1898 found that atoms could sometimes eject a far smaller negative particle which he called ELECTRON 1906 Nobel prize in Physics History of the atom 1808 John Dalton suggested that all matter was made up of tiny spheres that were able to bounce around with perfect elasticity and called them ATOMS History of the atom Democritusdevelopedthe idea of atoms 460 BC he pounded up materials in his pestle and mortar until he had reduced them to smaller and smaller particles which he called ATOMA (greek for indivisible) It took ~2400 years from when it was conceived to the time experimental evidence prove of the atom existence. properties of solidmaterials depend on thegeometrical atomicarrangements,and the interactions between constituent atoms. Understanding thestructure of atoms is critical to understanding the properties of matter. E N D - Presentation TranscriptĪtomic Structure All matter is composed of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
